Fight HCM with Felycin®-CA1
(sirolimus delayed-release tablets)
Felycin-CA1 is the first and only medication conditionally approved by the FDA for for the management of ventricular hypertrophy in cats with subclinical hypertrophic cardiomyopathy (HCM).
Now you can offer early intervention to your feline patients with subclinical HCM.
We studied it so you can prescribe it with confidence
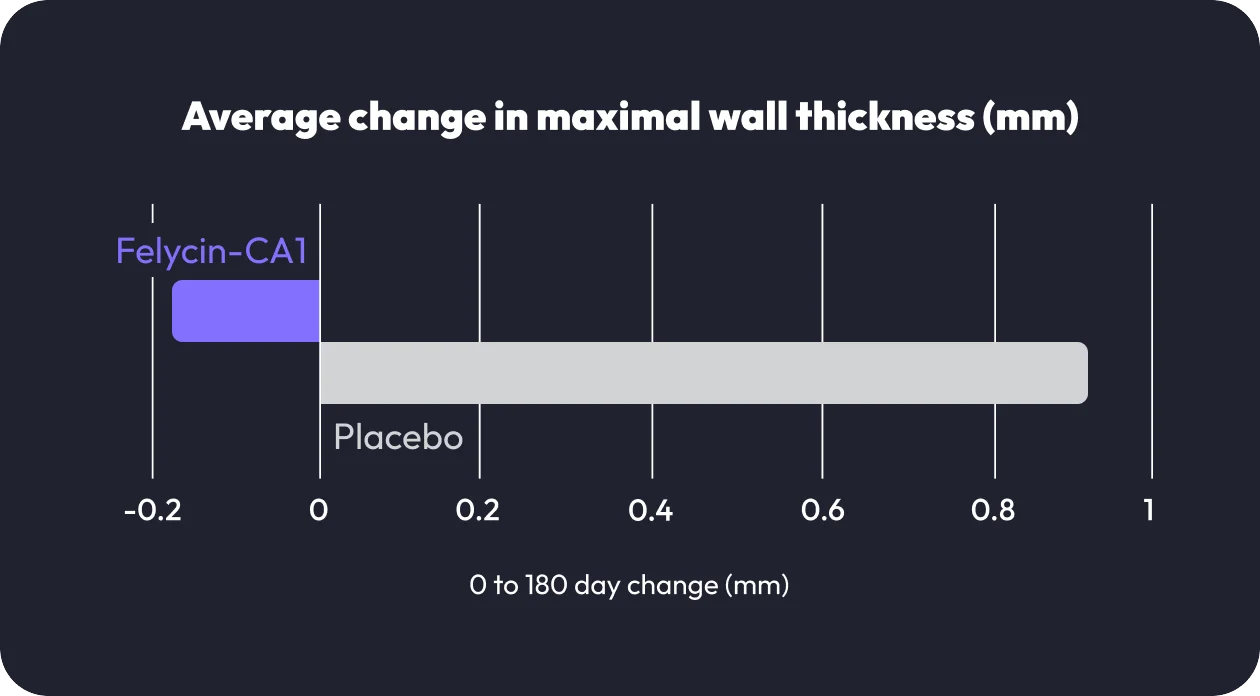
In the RAPACAT study, thirty-six cats with subclinical HCM were administered Felycin-CA1 or a placebo. Cats treated with Felycin-CA1 had a significantly lower left ventricular maximal wall thickness, with an average reduction of 0.17 mm in comparison to 0.94 mm increase for cats receiving placebo.1
The drug also underwent extensive safety studies, and Felcyin-CA1’s once-weekly dosing regimen demonstrated a favorable safety profile.
Felycin-CA1’s mode of action
What veterinarians are saying about Felycin-CA1

HCM: A new standard of care video

Clinically proven HCM Management Video

Hope for HCM Cats Video
Ready to prescribe Felycin-CA1?
Join the fight against HCM. To order Felycin-CA1, contact your local representative or authorized distributor.
Tools and Resources
Felycin-CA1 Detailer
Educate your team about subclinical HCM and Felycin-CA1.
Felycin-CA1 Pet Owner Brochure
Teach your clients about the signs of subclinical HCM and how Felycin-CA1 can help.
Felycin-CA1 Social Media Assets
Share these social media assets on your veterinary pages to spark conversations, raise awareness, and keep your clients informed.
If you’re not diagnosing HCM in 1 of every 7 cats you see, you may be missing a chance to intervene.
Your Experience Could Shape the Future of Feline Care
Have you treated a cat with Felycin-CA1? Share your success story and help other clinics see the difference early intervention can make.
Felycin-CA1 FAQ
References
1 Fuentes VL, Abbott J, Chetboul V, et al. ACVIM consensus statement guidelines for the classification, diagnosis, and management of cardiomyopathies in cats. JVIM 2020;34:1062-1077.
2 Kittleson MD, Cote E. The feline cardiomyopathies: Hypertrophic cardiomyopathy. JFMS 2021;23:1028-1051.
